Otto Hahn (who later discovered nuclear fission) worked under Rutherford at the Montreal Laboratory in 1905 - 1906. 
It was for this work that he was the Nobel Prize in Chemistry in 1908. He also noticed that a sample of radioactive material invariably took the same amount of time for half the sample to decay (known as its “ half-life”) and suggested a practical application using this constant rate of radioactive decay as a clock, which could then be used to help determine the age of the Earth (which turned out to be much older than most of the scientists at the time believed). His "disintegration theory" of radioactivity identified radioactive phenomena as atomic, not molecular, processes, due to the spontaneous disintegration of atoms. In 1900, he married Mary Georgina Newton, and they had a daughter, Eileen Mary, the next year.ĭuring his nine years in Montreal, Rutherford collaborated with the young Frederick Soddy (winner of the Nobel Prize in Chemistry in 1921) on ground-breaking research into the transmutation of elements. In 1898, Rutherford was appointed to the vacant chair of physics at McGill University in Montreal, Canada. 
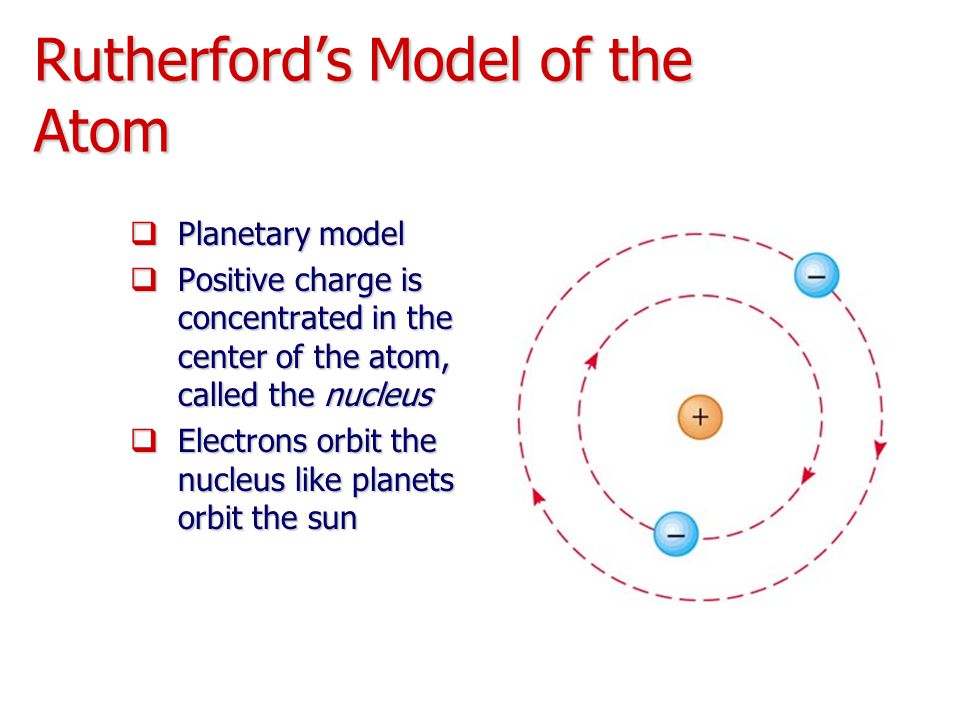
In 1897, he was awarded a BA Research Degree and the Coutts-Trotter Studentship of Trinity College, Cambridge. During Rutherford’s investigation of radioactivity at Cambridge, he invented an ingenious detector for electromagnetic waves, and coined the terms “ alpha” and “ beta” to describe the two distinct types of radiation emitted by thorium and uranium. Thompson (soon to become the discoverer of the electron). He continued with research work at Canterbury College for a short time, receiving a BSc degree in 1894, before traveling to England in 1895 for postgraduate study at the Cavendish Laboratory at the University of Cambridge, where he studied under J. He graduated with an MA in 1893, with a double first in Mathematics and Physical Science. He was educated at Havelock School and then, at age 16, Nelson Collegiate School, before winning a scholarship to study at Canterbury College at the University of New Zealand in Wellington in 1889. He was awarded the Nobel Prize in Chemistry in 1908 “for his investigations into the disintegration of the elements, and the chemistry of radioactive substances”.Įrnest Rutherford was born on 30 August 1871 in Spring Grove (now called Brightwater) near Nelson, New Zealand, the fourth of twelve children of a Scottish farmer and an English schoolteacher. He is also credited with the discovery of the proton in 1919, and hypothesized the existence of the neutron. In 1911, he was the first to discover that atoms have a small charged nucleus surrounded by largely empty space, and are circled by tiny electrons, which became known as the Rutherford model (or planetary model) of the atom. In contrast, the particles that were highly deflected must have experienced a tremendously powerful force within the atom.Ernest Rutherford, 1st Baron Rutherford of Nelson was a New Zealand chemist who has become known as the “father of nuclear physics”. Because the vast majority of the alpha particles had passed through the gold, he reasoned that most of the atom was empty space. Rutherford needed to come up with an entirely new model of the atom in order to explain his results. In a famous quote, Rutherford exclaimed that it was "as if you had fired a 15-inch shell at a piece of tissue and it came back and hit you." No prior knowledge had prepared them for this discovery. Some were even redirected back toward the source. Surprisingly, while most of the alpha particles were indeed not deflected, a very small percentage (about 1 in 8000 particles) bounced off the gold foil at very large angles. Rutherford found that a small percentage of alpha particles were deflected at large angles, which could be explained by an atom with a very small, dense, positively-charged nucleus at its center (bottom).Īccording to the accepted atomic model, in which an atom's mass and charge are uniformly distributed throughout the atom, the scientists expected that all of the alpha particles would pass through the gold foil with only a slight deflection or none at all. (B) According to the plum pudding model (top), all of the alpha particles should have passed through the gold foil with little or no deflection. \): (A) The experimental setup for Rutherford's gold foil experiment: A radioactive element that emitted alpha particles was directed toward a thin sheet of gold foil that was surrounded by a screen which would allow detection of the deflected particles.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
